Achillea millefolium (flower)
Contents |
Nomenclature
Achillea millefolium L. Asteraceae
Standardized common name (English): yarrow
Botanical Voucher Specimen
 |
|
|
|
|
[[Category:Tropicos.org. Missouri Botanical Garden. 05 Aug 2013 <http://www.tropicos.org/Image/88582>]]
Organoleptic Characteristics
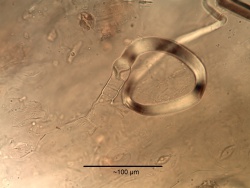
Macroscopic Characteristics
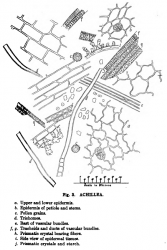
Microscopic Characteristics
|
High Performance Thin Layer Chromatographic Identification
|
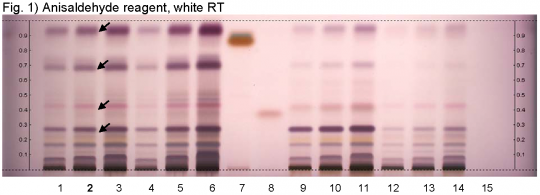
Yarrow (flower) (Achillea millefolium) Lane Assignments Lanes, from left to right (Track, Volume, Sample):
Reference Sample(s) Reference: Dissolve 25 mg of cineole in 20 mL of tolulene. Dissolve 10 mg of guaiazulene in 20 mL of tolulene. Stationary Phase Stationary phase, i.e. Silica gel 60, F254 Mobile Phase Tolulene, ethyl acetate 95:5 (v/v) Sample Preparation Method Sample: Mix 1.0 g of powdered sample with 10 mL of methanol and sonicate for 10 minutes, then centrifuge or filter the solutions and use the supernatants / filtrates as test solutions. Derivatization reagent: Anisaldehyde reagent; Preparation: 170 mL of ice cooled methanol are mixed with 20 mL of acetic acid, 10 mL of sulfuric acid, and 1 mL of anisaldehyde. Use: Dip (time 0, speed 5), heat at 100°C for 4 min. Detection Method Saturated chamber; developing distance 70 mm from lower edge; relative humidity 33% Other Notes Images presented in this entry are examples and are not intended to be used as a bases for setting specifications for quality control purposes. System suitability test: Cineole: violet zone at Rf ~ 0.38. Guaiazulene: brown zone at Rf ~ 0.88. Application: 2 µL of references, 16 µL of test solutions Identification: Compare result with reference images. The fingerprint of the test solution is similar to that of the corresponding botanical reference sample. Additional weak zones may be present. The chromatogram of the test solution shows a violet zone at Rf ~ 0.94 right above the zone due to guaiazulene reference substance. Below this zone there is a violet zone at Rf ~ 0.70. There is a reddish-violet zone at Rf ~ 0.43 right above the zone due to cineole reference substance. Below this zone there is a violet zone at Rf ~ 0.29 (black arrows).
|
Supplementary Information
Other Publications
2D NMR Barcoding and Differential Analysis of Complex Mixtures for Chemical Identification: The Actaea Triterpenes, Qiu et al.
The interpretation of NMR spectroscopic information for structure elucidation involves decoding of complex resonance patterns that contain valuable molecular information (δ and J), which is not readily accessible otherwise. We introduce a new concept of 2D-NMR barcoding that uses clusters of fingerprint signals and their spatial relationships in the δ−δ coordinate space to facilitate the chemical identification of complex mixtures. Similar to widely used general barcoding technology, the structural information of individual compounds is encoded as a specifics pattern of their C,H correlation signals. Software-based recognition of these patterns enables the structural identification of the compounds and their discrimination in mixtures. Using the triterpenes from various Actaea (syn. Cimicifuga) species as a test case, heteronuclear multiple-bond correlation (HMBC) barcodes were generated on the basis of their structural subtypes from a statistical investigation of their δH and δC data in the literature. These reference barcodes allowed in silico identification of known triterpenes in enriched fractions obtained from an extract of A. racemosa (black cohosh). After dereplication, a differential analysis of heteronuclear single-quantum correlation (HSQC) spectra even allowed for the discovery of a new triterpene. The 2D barcoding concept has potential application in a natural product discovery project, allowing for the rapid dereplication of known compounds and as a tool in the search for structural novelty within compound classes with established barcodes.[7]
Sources
- ↑ Tropicos.org. Missouri Botanical Garden. 05 Aug 2013 <http://www.tropicos.org/Image/88582> http://www.tropicos.org/Image/88582
- ↑ Botanical Voucher Specimen Library, Alkemists Laboratories http://www.alkemist.com http://www.alkemist.com
- ↑ Schneider, A. (1921) The Microanalysis of Powdered Vegetable Drugs, 2nd ed.
- ↑ Schneider, A. (1921) The Microanalysis of Powdered Vegetable Drugs, 2nd ed.
- ↑ Elan M. Sudberg, Alkemist Laboratories http://www.alkemist.com http://www.alkemist.com
- ↑ HPTLC Association http://www.hptlc-association.org/
- ↑ 2D NMR Barcoding and Differential Analysis of Complex Mixtures for Chemical Identification: The Actaea Triterpenes Feng Qiu, James B. McAlpine, David C. Lankin, Ian Burton, Tobias Karakach, Shao-Nong Chen, and Guido F. Pauli Analytical Chemistry 2014 86 (8), 3964-3972. http://pubs.acs.org/doi/abs/10.1021/ac500188j