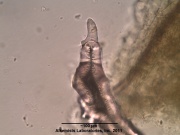
Camellia sinensis (leaf)
Contents |
Introduction
Introduction from Wikipedia, the free encyclopedia (http://en.wikipedia.org/wiki/Camellia_sinensis, retrieved 02/20/2012).
Camellia sinensis is the species of plant whose leaves and leaf buds are used to produce Chinese tea. It is of the genus Camellia (Chinese: 茶花; pinyin: Cháhuā), a genus of flowering plants in the family Theaceae. White tea, green tea, oolong, pu-erh tea and black tea are all harvested from this species, but are processed differently to attain different levels of oxidation. Kukicha (twig tea) is also harvested from Camellia sinensis, but uses twigs and stems rather than leaves. Common names include tea plant, tea tree, and tea shrub.
There are two major varieties used for tea, Chinese tea, Camellia sinensis var. sinensis, and Assam tea, Camellia sinensis var. assamica.
The quoted text in this section was licensed for use under the Creative Commons ShareAlike License, version 3.0: http://creativecommons.org/licenses/by-sa/3.0/
Macroscopic Entries
|
|
Microscopic Entries
|
|
|
HPTLC Entries
|
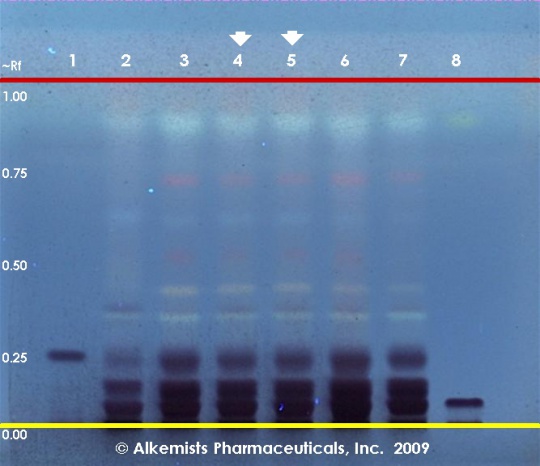
Green Tea (leaf) (Camellia sinensis) Lane Assignments Lanes, from left to right (Track, Volume, Sample):
Reference materials used here have been authenticated by macroscopic, microscopic &/or TLC studies according to the reference source cited below held at Alkemists Laboratories, Costa Mesa, CA. Stationary Phase Silica gel 60, F254, 10 x 10 cm HPTLC plates Mobile Phase CHCl3: ethyl formate: HCOOH [5/4/1] Sample Preparation Method 0.3 g + 3ml 70% grain EtOH sonicated + heated @ 50° C ~ 1 hr Detection Method Vanillin/H2SO4 Reagent -> 110° C 5 min -> UV 365 nm Reference see Herbal Drugs and Phytopharmaceuticals, Wichtl, M., 1994
|
|
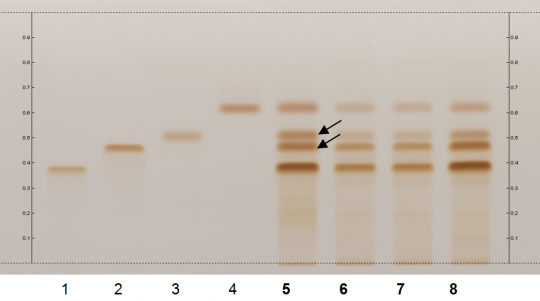
Green Tea (leaf) (Camellia sinensis) Lane Assignments Lanes, from left to right (Track, Volume, Sample):
Reference Sample(s) Reference:Individually dissolve 1 mg of (-)-epigallocatechin and 1 mg of (-)-epicatechin gallate each in 20 mL of methanol; Optional: Individually dissolve 1 mg of (-)-epigallo-catechin-3-O-gallate and 1 mg of (-)-epicatechin each in 20 mL of methanol; Store all solutions at -20°C. Stationary Phase Stationary phase, i.e. Silica gel 60, F254 Mobile Phase Toluene, acetone, formic acid 9:9:2 (v/v/v) Sample Preparation Method Sample: Mix 100 mg of powdered sample with 10 mL of methanol, water 4:1 and sonicate for 10 minutes, then centrifuge or filter the solutions and use the supernatants / filtrates as test solutions. Derivatization reagent: Fast Blue salt B reagent; Preparation: dissolve 140 mg of Fast Blue salt B in 10 mL of water and add 140 mL of methanol and 50 mL of dichloromethane. Store reagent in the dark at 4°C; Use: preheat the plate to 100°C for 2 min, then dip (time 0, speed 5), dry for 5 min in the fume hood. Detection Method Unsaturated chamber; developing distance 60 mm from lower edge; relative humidity 33% Other Notes Images presented in this entry are examples and are not intended to be used as basis for setting specifications for quality control purposes. System suitability test: (-)-Epigallocatechin: brown zone at Rf ~ 0.46; (-)-Epicatechin gallate: brown zone at Rf ~ 0.52 Identification: Compare result with reference images. The fingerprint of the test solution is similar to that of the corresponding botanical reference sample. Additional weak zones may be present. The chromatogram of the test solution shows four brownish-orange zones corresponding to reference substance epigallocatechin-3-O-gallate (Rf ~ 0.37), (-)-epigallocatechin (Rf ~ 0.46), (-)-epicatechin gallate (Rf ~ 0.52), and (-)-epicatechin (Rf ~ 0.62). The lowest zone is the most intense and the upper zone is the faintest. The two zones in between are clearly separated (black arrows).
|
Other Points of Interest
Cite error: <ref> tags exist, but no <references/> tag was found