Nomenclature
Carthamus tinctorius L. Asteraceae
Standardized common name (English): safflower
Ayurvedic name(s): kusumbha
Pinyin name(s): hong hua (flower)
Botanical Voucher Specimen
Organoleptic Characteristics
Macroscopic Characteristics
| Carthamus tinctorius L. Safflower.—
The African, false, American, or dyers' saffron is an annual composite, with a smooth, erect stem, somewhat branched at top, and a foot or two in height. ... The florets are in mass of a red color, diversified by the yellow of the styles contained within the floret. It has a peculiar, slightly aromatic odor, and a scarcely perceptible bitterness.
It contains a fixed oil; also two coloring substances—one red, insoluble in alkaline liquids, and called carthamin or carthamic acid by Dobereiner, who found it to possess weak acid properties; the other yellow, and soluble in water.
Source: United States Dispensatory (1918) [1]
|
|
|
|
Microscopic Characteristics
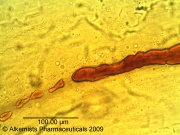
| Fragment of the corolla showing brown laticiferous vessels observed at 400x with Acidified Chloral Hydrate Glycerol Solution.cellular structures identified in this botanical specimen are fragment of the corolla showing brown laticiferous vessels and three pored pollen grain shown exuding contents when observed at 400x with Acidified Chloral Hydrate Glycerol Solution.
Source: Elan M. Sudberg, Alkemist Laboratories [2]
|
|
|
|
High Performance Thin Layer Chromatographic Identification
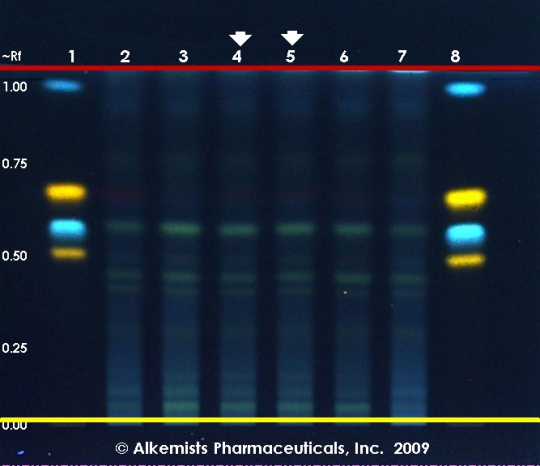
Carthamus tinctorius HPTLC ID - Natural Product Reagent + PEG UV 365 nm
Safflower (flower) (Carthamus tinctorius)
Lane Assignments Lanes, from left to right (Track, Volume, Sample):
- 1 μL Rutin, Caffeic Acid, Hyperoside, Chlorogenic Acid ~ 0.1% in Methanol
- 3 μL Carthamus tinctorius-1 (flower)
- 3 μL Carthamus tinctorius-2 (flower)
- 3 μL Carthamus tinctorius-3 (flower)
- 3 μL Carthamus tinctorius-3 (flower)
- 3 μL Carthamus tinctorius-4 (flower)
- 3 μL Carthamus tinctorius-5 (flower)
- 1 μL Rutin, Caffeic Acid, Hyperoside, Chlorogenic Acid ~ 0.1% in Methanol
Reference materials used here have been authenticated by macroscopic, microscopic &/or TLC studies according to the reference source cited below held at Alkemists Laboratories, Costa Mesa, CA.
Stationary Phase Silica gel 60, F254, 10 x 10 cm HPTLC plates
Mobile Phase ethyl acetate: AcCOOH: HCOOH: H2O [10/1.1/1.1/2.4]
Sample Preparation Method 0.3 g + 3 ml CH3OH sonicated + heated @ 50° C ~ 1 hr
Detection Method Natural Product Reagent + PEG -> UV 365 nm
Reference see Adapted from Plant Drug Analysis, Wagner, H., 1996
Source: Elan M. Sudberg, Alkemist Laboratories [3]
|
Supplementary Information
Sources
- ↑ United States Dispensatory (1918)
- ↑ Elan M. Sudberg, Alkemist Laboratories http://www.Alkemist.com
- ↑ Elan M. Sudberg, Alkemist Laboratories http://www.alkemist.com