Withania somnifera (root)
(HPTLC) |
|||
| Line 101: | Line 101: | ||
|} | |} | ||
=High Performance Thin Layer Chromatographic Identification= | =High Performance Thin Layer Chromatographic Identification= | ||
| + | {{HPTLC | source=HPTLC Association | ||
| + | | companyimage=HPTLC-assoc-Logo-farbig-Text-schwarz-300x47.png | ||
| + | | companyURL=http://www.hptlc-association.org/ | ||
| + | | mainimage=Withania somnifera - HPTLC ASSOC.png | ||
| + | | caption1= | ||
| + | | description=Ashwagandha (root) (''Withania somnifera'') | ||
| + | | | ||
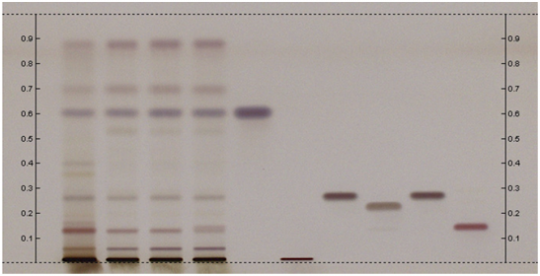
| + | | lanes=Lanes, from left to right (Track, Volume, Sample): | ||
| + | # 5 µL ''Withania somnifera'' extract (20 mg/mL) | ||
| + | # 10 µL ''Withania somnifera'' root | ||
| + | # 10 µL ''Withania somnifera'' root | ||
| + | # 10 µL ''Withania somnifera'' root | ||
| + | # 2 µL β-Sitosterol | ||
| + | # 2 µL Withanoside IV | ||
| + | # 2 µL Withanolide A | ||
| + | # 2 µL Withanone | ||
| + | # 2 µL Withanolide D | ||
| + | # 2 µL Withaferin A | ||
| + | | notes=<br> | ||
| + | '''Reference Standard Solution:'''0.2 mg/mL β-Sitosterolin methanol and 0.2 mg/mL withanolide A in methanol. Optional: 0.2 mg/mL withaferin A in methanol. | ||
| + | '''Reference Sample Preparations:''' Sonicate 0.5g of powdered sample in 5mL of methanol for 15 minutes, centrifuge or filter the solution, and use the supernatant / filtrate. | ||
| + | |||
| + | '''Stationary Phase:''' HPTLC, Silica gel 60 F254 | ||
| + | |||
| + | '''Mobile Phase:''' Toluene, ethyl acetate, acetic acid (55:45:3) | ||
| + | |||
| + | '''Development:''' Saturated chamber, developing distance 70 mm from lower edge of the plate; relative humidity 33%, temperature 25°C. | ||
| + | |||
| + | '''Derivatization reagent:''' Sulfuric Acid Reagent– 10% sulfuric acid in methanol (prepare fresh) | ||
| + | |||
| + | '''Detection:''' Dip (time 0, speed 5) in Derivatization reagent, heat plate at 100°C for 5 min,and examine under visible light. | ||
| + | |||
| + | '''Procedure:''' | ||
| + | |||
| + | Reference Standard Solutions, Stationary Phase, Mobile Phase, Derivatization reagent, and Detection, as described above. | ||
| + | |||
| + | '''Test Sample Preparation:''' Prepare test sample as described under Reference Sample Preparations andapply 10uL. | ||
| + | |||
| + | '''Identification:''' Compare Test Sample Preparation chromatogram with chromatograms of Reference Sample Preparations. The Test SamplePreparation chromatogram is similar to that of the Reference Sample Preparations chromatograms. Additional weak zones may be present. | ||
| + | |||
| + | The Test Sample Preparation chromatogram exhibits a grey-violet zoneand a brownish-violet zone at Rf corresponding to those of β-Sitosterol and withanolide A, respectively, in Reference Standard Solutionchromatogram, and two browninsh-violet zones in the lower third of the chromatogram at Rf lower than that corresponding to withanolide A in Reference Standard Solution chromatogram. | ||
| + | |||
| + | | }} | ||
=Supplementary Information= | =Supplementary Information= | ||
Revision as of 22:06, 6 November 2014
Contents |
Nomenclature
Withania somnifera (L.) Dunal Solanaceae
Standardized common name (English): ashwagandha
Ayurvedic name(s): ashvagandha
Botanical Voucher Specimen
|
|
Organoleptic Characteristics
|
Macroscopic Characteristics
|
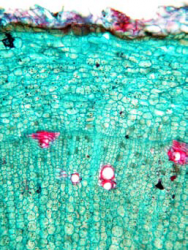
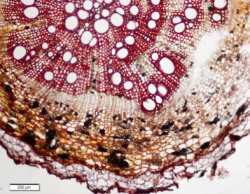
Microscopic Characteristics
|
High Performance Thin Layer Chromatographic Identification
|
Ashwagandha (root) (Withania somnifera) Lane Assignments Lanes, from left to right (Track, Volume, Sample):
Other Notes Reference Sample Preparations: Sonicate 0.5g of powdered sample in 5mL of methanol for 15 minutes, centrifuge or filter the solution, and use the supernatant / filtrate. Stationary Phase: HPTLC, Silica gel 60 F254 Mobile Phase: Toluene, ethyl acetate, acetic acid (55:45:3) Development: Saturated chamber, developing distance 70 mm from lower edge of the plate; relative humidity 33%, temperature 25°C. Derivatization reagent: Sulfuric Acid Reagent– 10% sulfuric acid in methanol (prepare fresh) Detection: Dip (time 0, speed 5) in Derivatization reagent, heat plate at 100°C for 5 min,and examine under visible light. Procedure: Reference Standard Solutions, Stationary Phase, Mobile Phase, Derivatization reagent, and Detection, as described above. Test Sample Preparation: Prepare test sample as described under Reference Sample Preparations andapply 10uL. Identification: Compare Test Sample Preparation chromatogram with chromatograms of Reference Sample Preparations. The Test SamplePreparation chromatogram is similar to that of the Reference Sample Preparations chromatograms. Additional weak zones may be present. The Test Sample Preparation chromatogram exhibits a grey-violet zoneand a brownish-violet zone at Rf corresponding to those of β-Sitosterol and withanolide A, respectively, in Reference Standard Solutionchromatogram, and two browninsh-violet zones in the lower third of the chromatogram at Rf lower than that corresponding to withanolide A in Reference Standard Solution chromatogram.
|
Supplementary Information
Sources
- ↑ MOBOT, Tropicos.org http://www.tropicos.org/Image/100184298
- ↑ Natural Remedies Pvt Ltd http://www.naturalremedy.com/
- ↑ Natural Remedies Pvt Ltd http://www.naturalremedy.com/
- ↑ Encyclopedia of Life http://eol.org/data_objects/19246862
- ↑ Encyclopedia of Life http://eol.org/data_objects/19246863
- ↑ Encyclopedia of Life http://eol.org/data_objects/19246864
- ↑ Natural Remedies Pvt Ltd http://www.naturalremedy.com/
- ↑ Natural Remedies Pvt Ltd http://www.naturalremedy.com/
- ↑ Natural Remedies Pvt Ltd http://www.naturalremedy.com/
- ↑ Natural Remedies Pvt Ltd http://www.naturalremedy.com/
- ↑ Natural Remedies Pvt Ltd http://www.naturalremedy.com/
- ↑ HPTLC Association http://www.hptlc-association.org/